(In fact, it’s all virtually gone within 50,000 years). Because they are millions of years old, they contain no carbon-14. The problem with fossil fuelsįossil fuels such as coal and oil come from ancient organisms. But new analyses show its usefulness is ending far earlier than it would have without recent growing emissions of carbon-based pollutants due to widespread use of fossil fuels. 
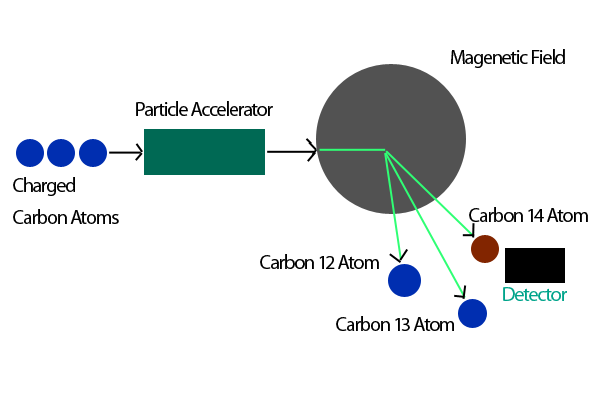
As carbon cycles through living things, the share of this isotope would naturally fall over time. Scientists knew that the fallout’s carbon-14 signal wouldn’t last forever. This ratio made carbon dating suitable for analyzing artwork, samples of tea, an unidentified body - or even a tusk of elephant ivory found in the back of a truck. Michael MacArthur/Harvard Medical School ( SITN Boston) ( CC BY-NC-SA 4.0) The slope of the line from the 1930s - showing atmospheric carbon-14 levels - would have remained low if it weren’t for the weapons tests. The spike is the pulse, or ‘bomb curve,’ due to nuclear weapons tests. This graph shows Earth’s changing carbon-14 levels since 1930. The black line shows scientists’ observed data. Now, instead of using the natural decay of carbon-14 to date things, scientists now could use a change in the ratio of carbon-14 to stable carbon-12. After the tests, there was enough carbon-14 in recent things to be able to measure. The sudden burst of extra carbon-14 from those bomb tests gave scientists a bookmark in time. A well-known graph of this has been nicknamed the “bomb curve.” It was like having a fresh source of carbon-14. (Thankfully, these tests ended in 1963.) Fallout from those nuclear bombs suddenly - and dramatically - boosted the amount of carbon-14 on or near Earth’s surface. military carried out a large number of above-ground nuclear weapons tests. Explainer: Radioactive dating helps solve mysteriesīut all that changed during the middle of the last century. Not enough of their carbon-14 had decayed to be easily measured. Scientists can figure out about how old a material is based on how much of that carbon-14 is left.Īt first, this technique was useful only for dating fairly old artifacts - items maybe 10,000 to 50,000 years old. This natural ratio of isotopes (carbon-12 to -13 to -14) had stayed fairly constant over geologic time. The isotope used for carbon dating - carbon-14, which has eight neutrons - accounts for only one atom in a trillion. Another 1.1 percent is carbon-13, which has seven neutrons. Some 98.9 percent exists as carbon-12, which has six protons and six neutrons. It takes 5,730 years for their levels to drop by 50 percent. After death, amounts of carbon-14 gradually start to fall as the radioactive atoms in their once-living tissues begin to decay. While organisms are alive, the carbon cycle ensures they all have about the same level of carbon-14 in their cells. One widely used dating technique relies on the clock-like decay of carbon-14. Scientists can use several different elements to date objects from the past. They described the problem July 19 in the journal Nature. That’s the findings of an international team of scientists. Explainer: Radiation and radioactive decay The reason is society’s rampant burning of fossil fuels. But for modern artifacts, the use of this carbon dating has become a little less reliable. Scientists have been able to use that decay to figure out the age of once-living things up to 55,000 years old. This isotope is unstable, meaning it decays - morphs into another element over time. Most of it will be the stable form: carbon-12, which is non- radioactive. This element comes in several forms, or isotopes. Carbon is the basis of life on Earth it’s in the cells of every living thing.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
